 News Release
News Release
World’s largest radiology AI marketplace CARPL raises $6m to accelerate the adoption of AI in clinical workflows
CARPL
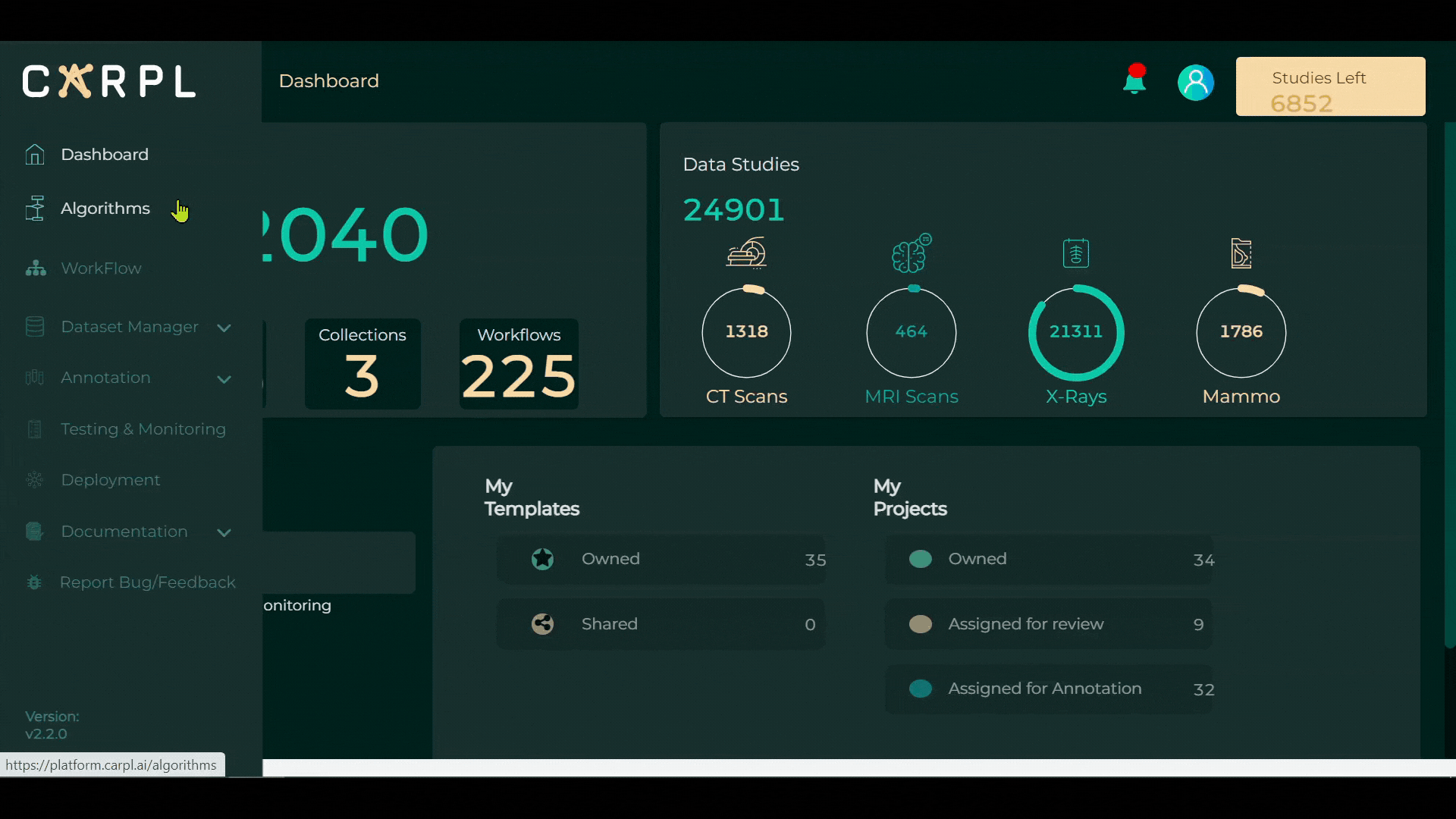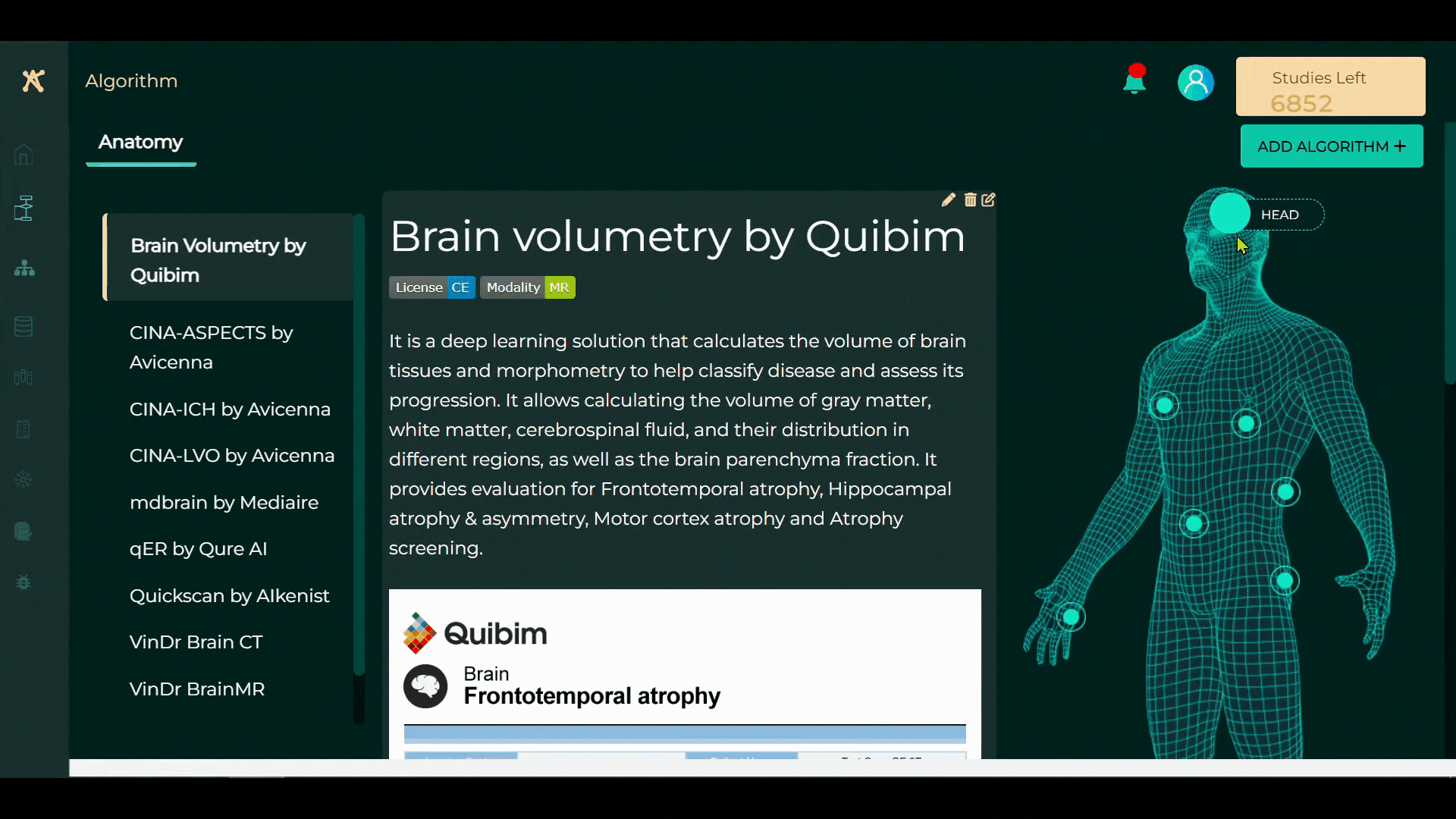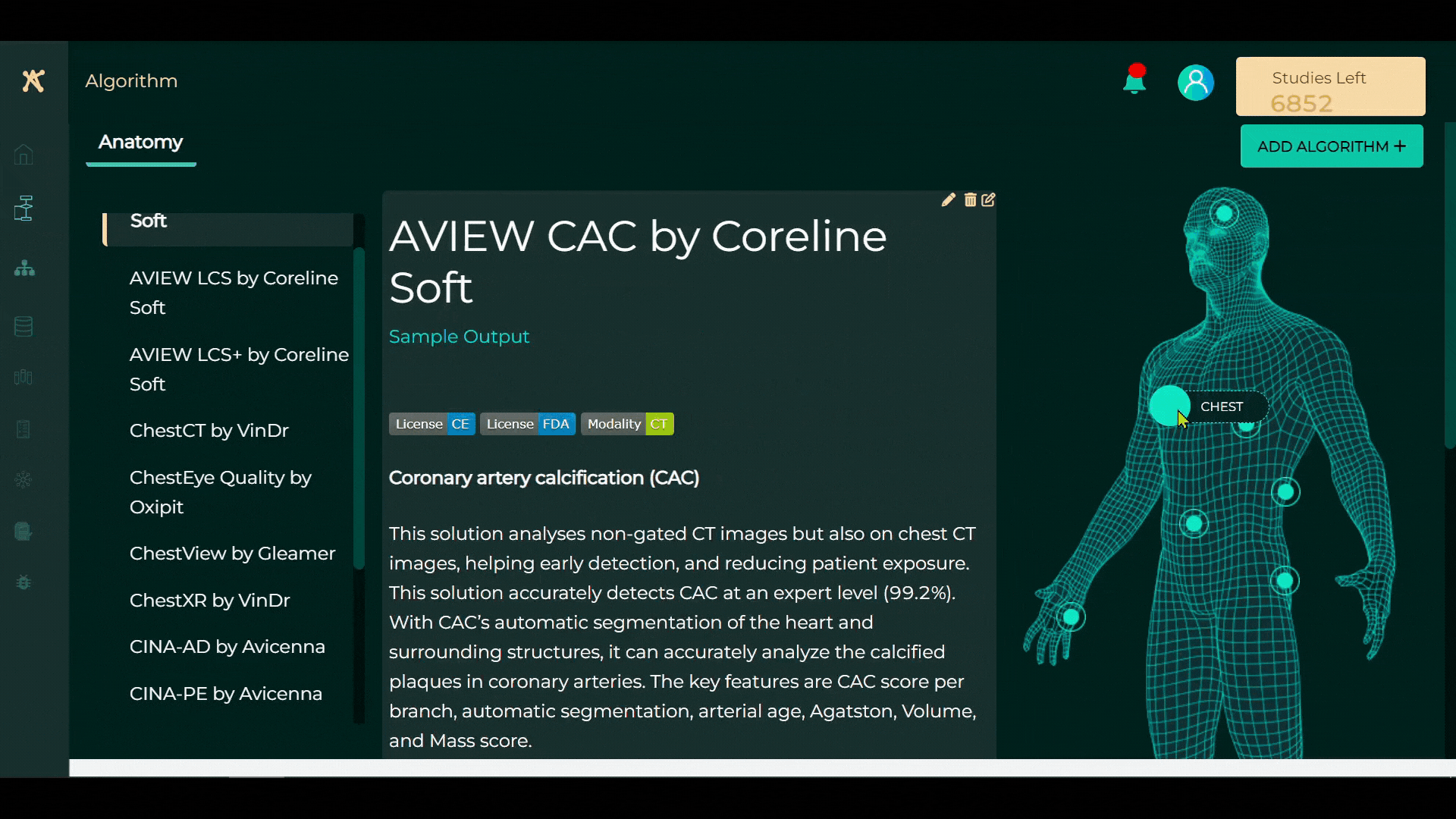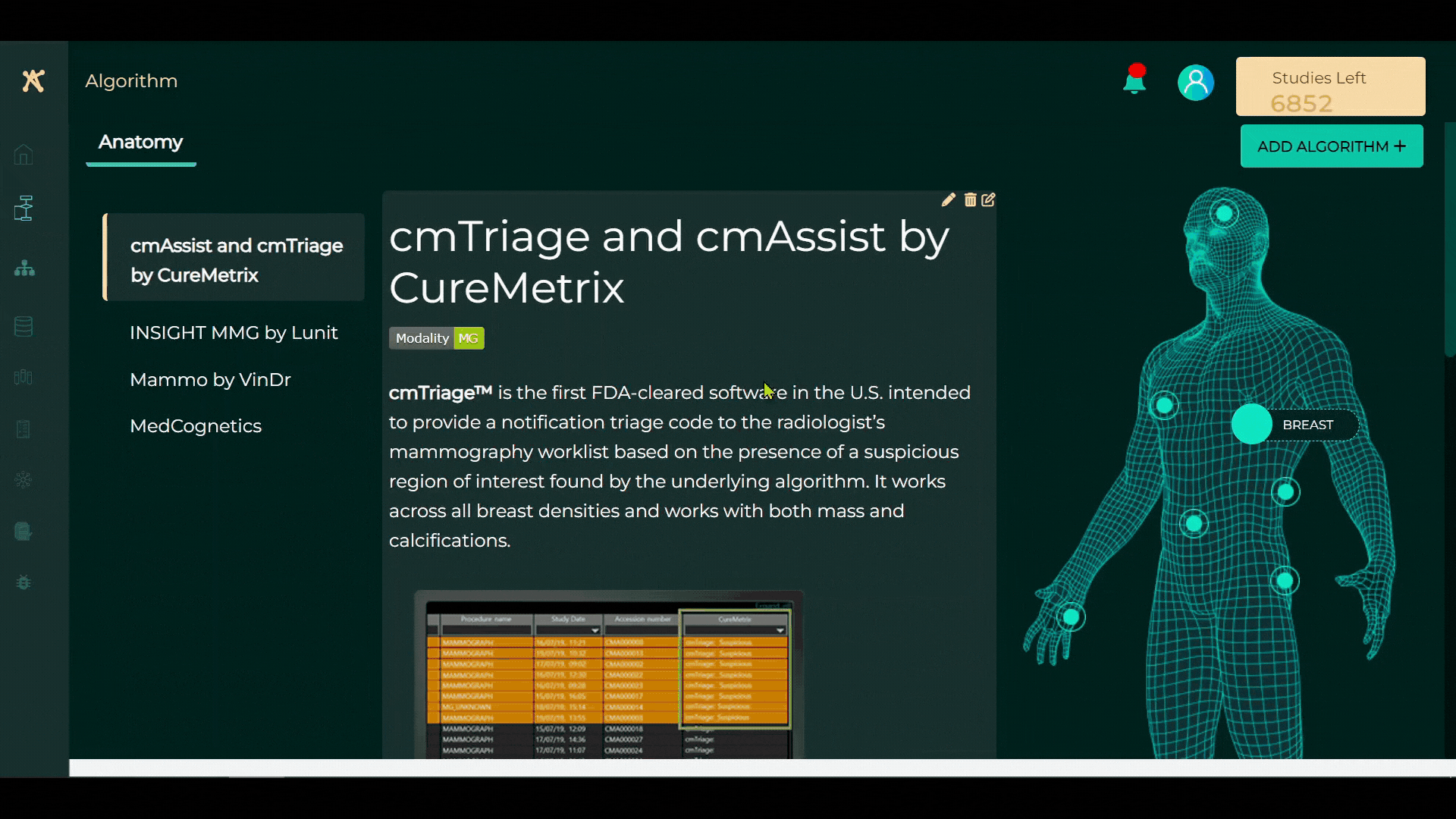
As people live longer and as emphasis on early detection of disease increases, there is a mounting burden on the healthcare services industry globally. This is being especially felt in radiology teams who are witnessing an acute shortage of radiologists. Currently, there are over 200 companies building applications using AI to automate parts of the radiologists’ work to bridge this talent gap. Today CARPL, an enterprise imaging AI marketplace platform, is announcing a $6m funding round to enable healthcare providers to access, assess and integrate these radiology AI applications into their clinical workflows through a single user-interface, single data channel and single procurement system. The seed funding round was led by Stellaris Venture Partners, a leading enterprise software investor, with participation from strategic angel investors* from Novo Holdings, Leapfrog PE, Bain & Co, Boston Consulting Group, UnitedHealth Group among others. CARPL will use the fresh funds to expand the team in North America and continue to build its tech stack. With more than 700 US-FDA approved AI applications, healthcare providers grapple with navigating the complex landscape of niche AI solutions. The daunting task involves determining the best AI solution for them and their patients, then seamlessly integrating it in the radiologists' workflows. This complexity has resulted in the sluggish pace of AI adoption in healthcare. A recent joint statement by the world’s top radiology associations also brings to light the importance of validation, deployment and monitoring of AI while being used in clinical practice. CARPL’s technology platform addresses this need using its proprietary DEV-D framework allowing healthcare providers to first Discover (D), Explore (E) and Validate (V) AI applications from CARPL’s AI marketplace, and subsequently Deploy (D) the most appropriate application across their clinical workflows. CARPL stands out as the only platform in the market with deep AI validation and monitoring capabilities, thereby ensuring safe and seamless AI deployment while upholding patient-centricity at its core. Additionally, CARPL’s universal AI viewer serves as a single user interface for radiologists, enabling them to accept, reject or refine AI outputs, which can subsequently be used to improve the performance of the AI itself. Operational since 2021, CARPL was founded by Vidur Mahajan, a physician with an MBA from the Wharton School of Business who has spent more than 12 years in the diagnostics industry. CARPL’s leadership comprises Dr Vasanth Venugopal as Chief Medical Officer, Rohit Takhar as CTO and most recently Dhruv Sahai as COO. CARPL has also ramped up its advisory team with the appointment of Dr Vijay Rao, SVP of Enterprise Imaging at Jefferson Health and a past-President of the Radiology Society of North America. “Over the past two years, we have onboarded more than 50 AI developers having 100+ AI applications, which made us the largest AI marketplace in terms of number of AI applications offered to customers. We are proud that some of the largest healthcare enterprises in the world have vetted our technology and trust us to be their partner in their AI and automation journeys.” said Dr Vidur Mahajan, CEO of CARPL. Drawing parallels with the lab medicine industry, Vidur Mahajan added: “We are democratizing radiology in the same way that automation and robotics revolutionized lab medicine, where a single pathologist signs out thousands of tests each day. We envision a future where CARPL creates ‘super-radiologists’ - AI-enabled radiologists who are ten times more productive than traditional radiologists.” Vidur Mahajan brings deep domain expertise having previously grown revenue 10x to $20m in 10 years at his family business Mahajan Imaging, a leading radiology service provider. He conceptualized CARPL under the guidance of his father Dr Harsh Mahajan, a pioneer in radiology, while working on validation and deployment of AI at Mahajan Imaging. CARPL comprises a multidisciplinary team with clinicians, scientists, engineers and business experts, all working towards the singular goal of driving exponential health outcomes using the encapsulated value of the entire AI ecosystem with the ultimate aim of ensuring access of quality diagnostics to patients. CARPL has more than 120 papers and conference presentations to its credit, and has coined some seminal concepts in clinical AI such as algorithmic audits, clinical explainability failure and unboxing AI. CARPL’s technology platform is used by the world’s top healthcare organizations like the Singapore Government, Massachusetts General Hospital (Boston), Radiology Partners (Los Angeles), University Hospitals (Cleveland), I-MED Radiology (Australia), Albert Einstein Hospital (São Paulo), Clinton Health Access Initiative, to name a few. CARPL is also creating impact in the public health space by working with the Government of India to enable large scale Tuberculosis Screening Programmes in the most remote regions of the country. As the global disease burden increases, owing to aging populations, early detection of disease and an increase in chronic/lifestyle diseases, the need for radiology services will increase, which increases the need for AI-assistance and automation in the delivery of these services. The global healthcare AI market is expected to cross $170 billion by 2029, of which almost $20 billion will be in the domain of radiology. Today, of the about 700 AI-based applications which are approved by the US-FDA, 80% are related to radiology. This number is expected to grow exponentially as access to training data and computation power becomes cheaper and easier to access. Alok Goyal, Partner at Stellaris Venture Partners commented: “The volume of imaging scans show a steady 9% year-on-year growth, outpacing the growth in the number of radiologists at a mere 1.8%. Bridging this demand gap is a crucial challenge for healthcare providers, and we believe AI will be the key. CARPL's integrated platform, designed for testing, deploying, and monitoring radiology AI applications, is poised to empower healthcare providers by seamlessly integrating AI into their clinical workflows. We are thrilled to join forces with Vidur and his team on this transformative journey.” As it becomes easier to build AI, the number and nature of AI applications used by doctors to deliver healthcare will increase exponentially. A platform approach that builds on connecting these AI developers to healthcare providers is the only way to exponentially improve the access, affordability and quality of care delivered today. CARPL is a new layer in the healthcare CIO’s stack that enables seamless access to the entire AI and analytics ecosystem through a single integrated platform. About CARPL CARPL is democratizing access to high quality healthcare by connecting AI applications and healthcare providers, exponentially improving the access, affordability and quality of medical care. CARPL enterprise imaging AI platform provides a single user-interface, data channel and procurement system for healthcare providers to access, assess, integrate and buy radiology AI applications in a safe and seamless manner. CARPL was incubated at Mahajan Imaging & Labs, India’s largest radiology service provider, as its technology division focused on building scalable clinical tools for AI validation, deployment and monitoring, under the guidance of Dr Harsh Mahajan, a pioneer in the field of radiology. For more information please visit https://carpl.ai About Stellaris Venture Partners Stellaris is an early-stage tech-focused VC firm that partners with fearless founders who dream big, think differently, and have an unstoppable desire to challenge the status quo. The fund works with these teams at nascent stages to help transform their ideas into resilient businesses. Stellaris does so with a team of seasoned, diverse operators and entrepreneurs who bring significant experience in building businesses and by providing access to some of the most successful founders and professionals. Since its inception in 2017, Stellaris has backed many market leaders such as Mamaearth, Whatfix, Propelld, Turno, Rigi, and others. For more information on Stellaris Venture Partners, visit https://www.stellarisvp.com Contact Details CARPL Bilal Mahmood +44 7714 007257 b.mahmood@stockwoodstrategy.com Company Website https://carpl.ai/
February 01, 2024 09:00 AM Eastern Standard Time
Image
 News Release
News Release